|
Which is element has a larger atomic radius than sulfur? The increased effective nuclear charge tends to decrease atomic radii across the Period from left to right. While aluminum has more electrons, valence electrons are known to shield each other very imperfectly. This means that the electrons are less attracted to the nucleus thus resulting in a larger size.ĭoes sodium or aluminum have a larger atomic radius? In phosphorus, there are fewer protons in the nucleus. This is due to the difference in the number of protons in the nucleus which affects the effective nuclear charge. Phosphorus has a larger atomic radius than Sulfur. Sulfur/Atomic radius Does P or S have a larger atomic radius? As a result, it is harder for sulfur atoms to come close enough together to form bonds. The radius of a sulfur atom is about 60% larger than that of an oxygen atom. Which has a larger atomic radius oxygen or sulfur? Atomic radius increases as you go to the left and downward due to the attraction of electrons and the nucleus in an atom. Which element has a larger atomic radius?Įxplanation: Francium has the largest, Helium has the lowest. Therefore, oxygen is expect to have a smaller atomic radius than of sulfur. Which elements have atomic radii smaller than sulfur?Įxplanation: Periodic trends indicate that atomic radius increases up a group and from left to right across a period. The farther these electrons are, the bigger the atom is. 
Silicon has an atomic number of 14 and, thus, has 14 protons. When neutral, sulfur should have 16 electrons as well. Sulfur has an atomic number of 16 which means that it has 16 protons. Why does silicon have a larger atomic radius than sulfur? 4 Which is element has the least attraction for an electron?. 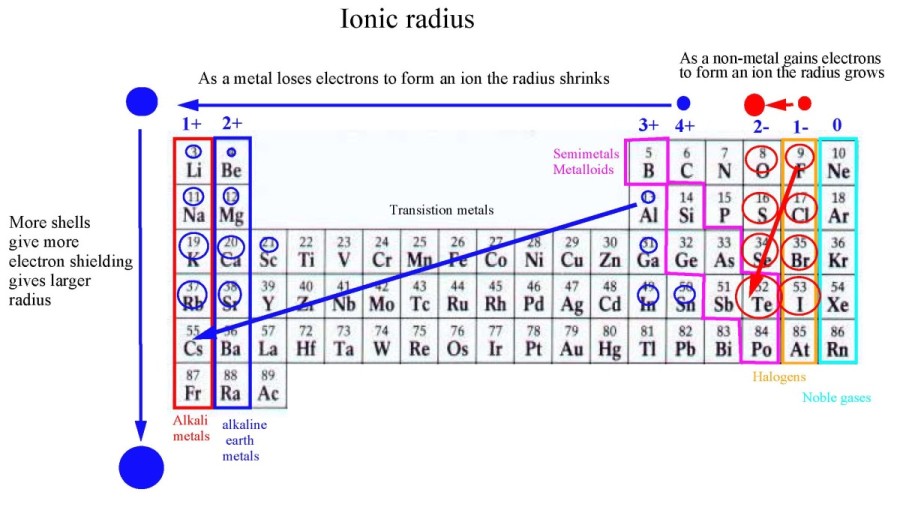
3 Does P or S have a larger atomic radius?.2 Which has a larger atomic radius oxygen or sulfur?.1 Why does silicon have a larger atomic radius than sulfur?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
